Medical devices demand digital traceability.
From UDI compliance to recall management, medical device companies need a digital connection to every device in the field. Veribl provides registration, documentation, service tracking, and safety communication — all from one QR code on the device.
Compliance & Traceability
Every device. A complete digital record.
UDI compliance, service history, calibration schedules — all tracked per device, audit-ready at any time.
Device compliance
3 devicesPulse Monitor PM-300
Warranty: active
Infusion Pump IV-44
Warranty: active
BP Cuff BC-120
Warranty: expired
All 3 devices UDI-verified and audit-ready
UDI compliance, automated.
Serialized QR codes encode device identification data. Every unit is traceable from manufacturing to clinical deployment — meeting FDA UDI and EU MDR requirements.
Audit-ready records, always.
Complete service history per device — calibrations, maintenance, field corrections. Ready for regulatory inspection at any time, with no manual record-keeping.
How it works
From registration to recall management.
One QR code connects every device in the field to its complete digital record.
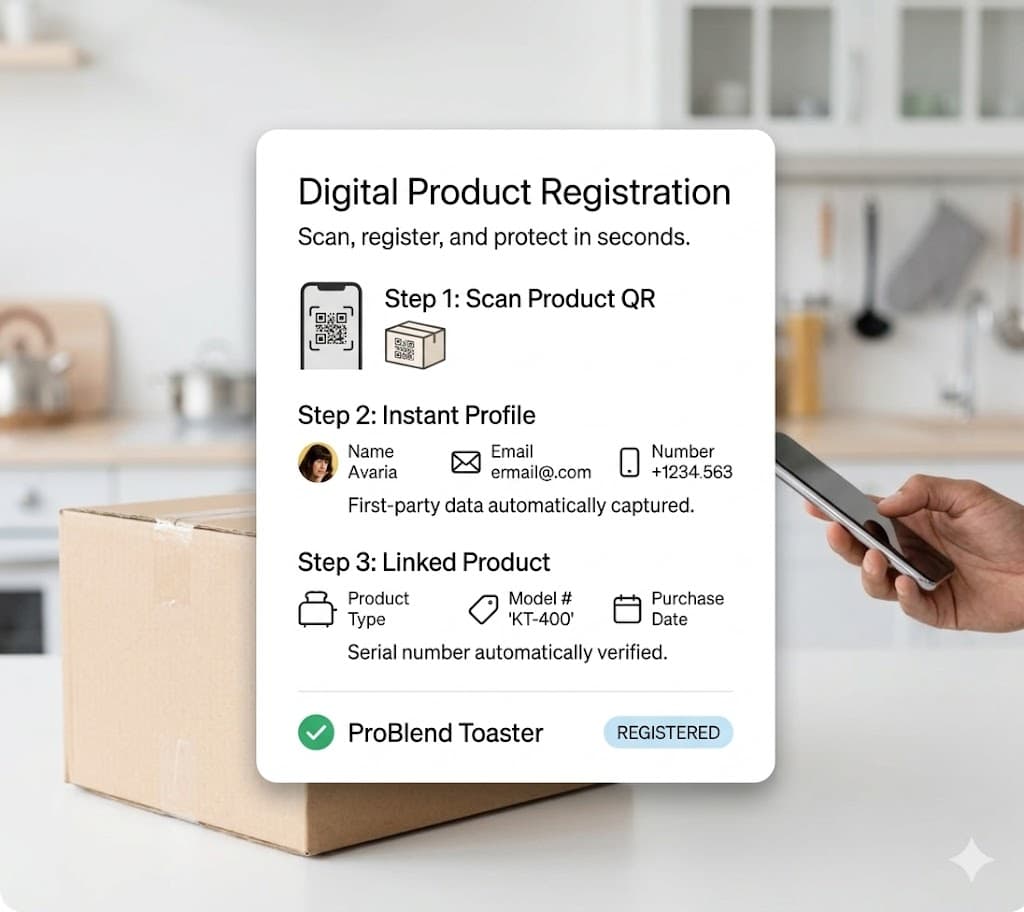
Device registration at point of use.
Healthcare professionals scan the QR on the device to register it at deployment. Veribl creates a complete traceability chain from manufacturing to clinical use — required by FDA UDI and EU MDR.
QR scan verified
Device registration
Device
Pulse Monitor PM-300
Serial number
PM300-22847
Registration
John Hopkins Medical — ICU Ward 3
UDI traceability chain established
Manufacturer → Distributor → Clinical deployment
Recall reach — 100% of registered devices.
When a recall is issued, Veribl identifies all affected devices and sends direct notifications to registered facilities. Track acknowledgment rates, automate follow-ups, and maintain a complete audit trail.
Complete platform
Built for medical device compliance.
Register your product
Activate your warranty and get support
Device registration & traceability.
QR-linked device identification. Each unit registered at point-of-use, creating the traceability chain regulators require.
Post-market surveillance data.
Real-world device performance data, usage patterns, and user-reported issues — the evidence regulators increasingly require for post-market surveillance.
Digital IFU & documentation.
Always-current digital Instructions for Use. When documentation updates, every device owner instantly accesses the latest version via QR scan.
See the platform in action
FAQ
Veribl provides a digital layer for medical devices that connects manufacturers to every device in the field. A QR code on each device links to registration, digital IFU, warranty status, service history, and safety communications. This improves UDI compliance, enables faster recalls, reduces IFU-related complaints, and provides post-market surveillance data — all from a single platform.
Yes. Veribl's serialized QR codes can encode or link to UDI data including Device Identifier (DI) and Production Identifier (PI). Each device's digital record maintains its full identification chain — manufacturer, model, lot/batch, serial number, and expiration date. This supports both FDA UDI and EU MDR requirements for device traceability.
When a recall is issued, Veribl identifies all registered devices matching the affected criteria (lot, serial range, date range) and sends direct notifications to registered owners or facilities. You get real-time tracking of recall acknowledgment rates, can send automated follow-ups to non-respondents, and maintain a complete audit trail for regulatory reporting.
Veribl can supplement or replace paper IFU with digital versions accessible via QR scan. Digital IFU can be updated instantly when documentation changes — no reprinting or redistribution required. This is particularly valuable for devices with frequent software updates or revised protocols. Note: check local regulations as some markets still require paper IFU availability.
Each device's digital record tracks its complete service history — calibrations, preventive maintenance, parts replacements, and field corrections. Automated alerts notify device owners when calibration or maintenance is due. Your service team can verify the full history before any intervention, and you maintain audit-ready records for regulatory inspections.
Yes. Veribl works across Class I, II, and III devices. The platform's flexibility allows configuration for different documentation requirements, warranty terms, and service schedules per device class. For Class III devices with more stringent post-market surveillance requirements, Veribl's data capture capabilities provide the real-world evidence regulators expect.
Turn every product into your best channel.
Get started in minutes, or book a personalized walkthrough with our team.